Unlocking the Power of Internal Audits: A Comprehensive Guide to מבדק פנימי
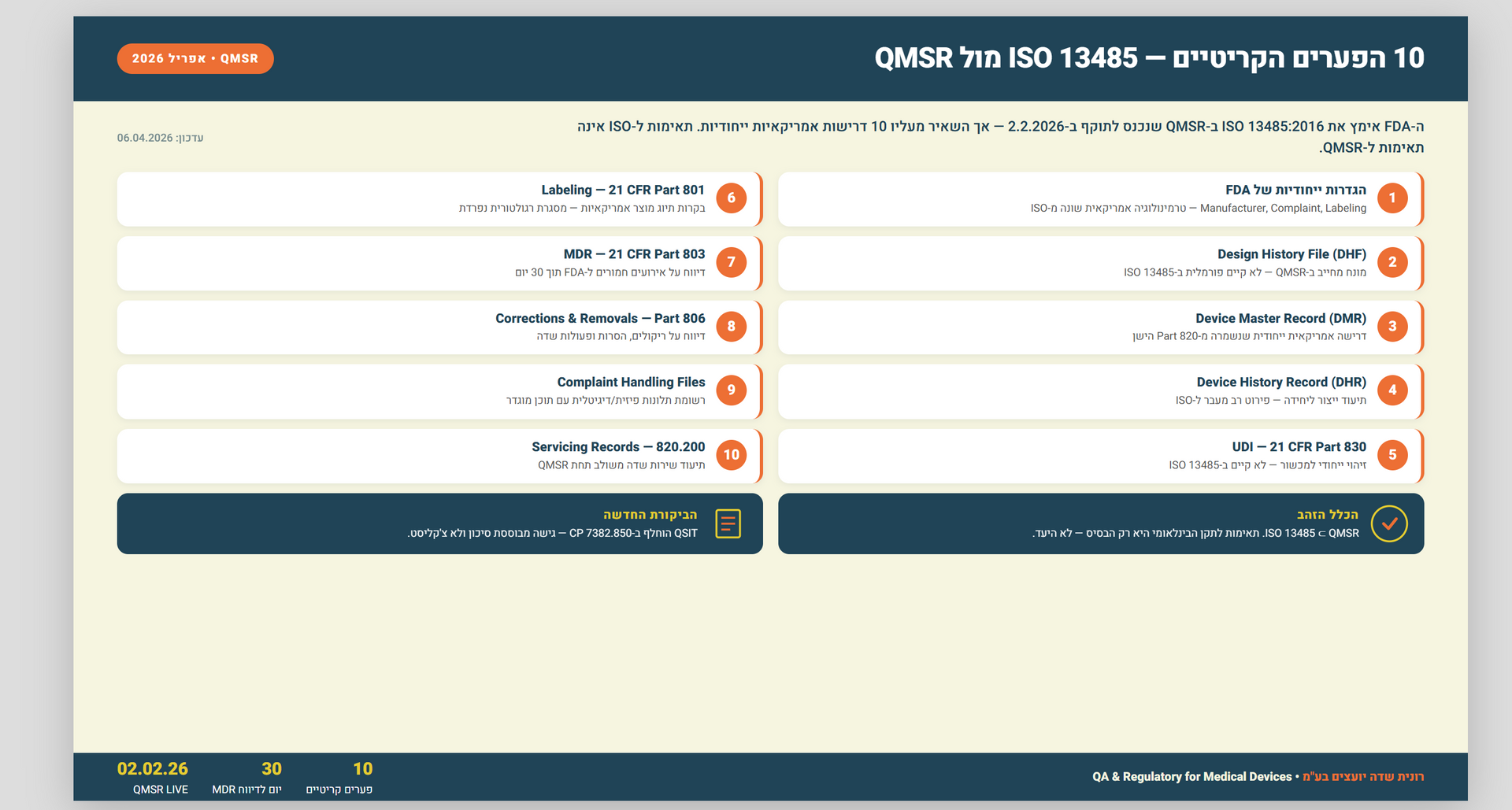
## Introduction to internal audits and their importance An internal audit is a vital element of an organization's risk management strategy. It offers a systematic and independent assessment of the organization's internal controls, processes, and procedures. The goal of these audits is to pinpoint potential risks, evaluate the effectiveness of internal controls, and ensure compliance with pertinent regulations and standards. Particularly for ISO certification, internal audits are critical in verifying an organization's adherence to the ISO standards' requirements. Understanding the concept of מבדק פנימי (internal audit) An internal audit, or מבדק פנימי in Hebrew, involves a systematic examination of an organization's internal controls, risk management practices, and compliance with relevant laws and regulations. This process includes a thorough review of the organization's operations, processes, and procedures to detect areas for enhancement, potential risks, and compliance shortfalls. The primary goal of an internal audit is to provide independent assurance to management and stakeholders that the organization's operations are efficient and effective, and in alignment with its strategic goals. Benefits of conducting מבדק פנימי Conducting an internal audit, or מבדק פנימי, delivers numerous advantages to organizations. It aids in the detection and mitigation of potential risks, bolstering risk management. Through regular internal audits, organizations can proactively strengthen their internal controls and reduce the chances of fraud, errors, or non-compliance. Moreover, internal audits empower organizations to uncover areas for operational efficiency enhancement, optimize resource use, and provide management with critical insights into their decision-making processes and the organization's overall performance. The role of מבדק פנימי in ISO certification For entities pursuing ISO certification, internal audits are indispensable in evaluating the organization's compliance with ISO standards. These audits, integral to the certification process, scrutinize the organization's adherence to the ISO standard's requirements. They assist in identifying any discrepancies or areas of non-conformity that must be rectified prior to certification. Additionally, internal audits offer a platform for continuous process and system improvement, ensuring alignment with ISO standards' best practices. Steps to perform a מבדק פנימי Executing a מבדק פנימי demands a systematic approach to guarantee thoroughness and accuracy. The steps below provide a framework for the internal audit process: Planning: Define the scope and objectives of the audit, establish a timeline, and identify the resources required. Preparation: Gather relevant documentation, such as policies, procedures, and previous audit reports. Familiarize yourself with the organization's processes and controls. Fieldwork: Conduct interviews with key personnel, observe operations, and review documentation to assess compliance, effectiveness, and efficiency. Analysis: Analyze the findings to identify areas for improvement, potential risks, and non-conformities. Reporting: Prepare a comprehensive audit report that includes the findings, recommendations, and action plans for addressing any identified issues. Follow-up: Monitor the implementation of the action plans and verify their effectiveness.
Common challenges in conducting מבדק פנימי and how to overcome them While conducting an internal audit can pose various challenges, proper planning and execution can surmount these obstacles. A common issue is the scarcity of resources, such as time and expertise. To tackle this, organizations might consider outsourcing their internal audit tasks to external consultants or enhancing the training of their internal auditors. Another hurdle is ensuring the independence and objectivity of the audit, which can be achieved by setting clear reporting structures and empowering auditors with the necessary authority and management support. Additionally, overcoming resistance to change and securing employee cooperation can be challenging, but emphasizing the audit's benefits and the critical role of employee participation can help alleviate these issues. Best practices for a successful מבדק פנימי איזו (internal audit process) To guarantee a successful internal audit process, organizations should adopt the following best practices: Establish a robust internal audit program: Develop a comprehensive plan that outlines the frequency, scope, and objectives of internal audits. Ensure independence and objectivity: Ensure auditors have the necessary independence, objectivity, and authority to conduct the audit effectively. Promote employee involvement: Encourage employees to actively participate in the audit process by providing feedback, suggestions, and sharing their concerns. Continuous improvement: Use the findings from internal audits as an opportunity for continuous improvement by implementing corrective actions and monitoring their effectiveness. Stay up-to-date with regulations and standards: Regularly update internal audit procedures to align with changing regulations and standards.
Tools and resources for מבדק פנימי Organizations have access to a plethora of tools and resources to facilitate effective internal audit processes. These resources include sophisticated audit software, comprehensive checklists, ready-to-use templates, and a variety of training materials. Audit software can enhance the efficiency of internal audits by automating tasks such as scheduling, data collection, and report generation. Checklists and templates offer a uniform approach to ensure that all critical areas are addressed during the audit. Furthermore, training materials, which can range from in-depth courses to informative webinars, are instrumental in empowering auditors with the skills and knowledge necessary for conducting internal audits with precision. Hiring a מבדק פנימי איזו (internal audit consultant) For those organizations that may not have the necessary internal resources or expertise to carry out an internal audit, engaging an external consultant can be an effective alternative. An adept internal audit consultant can deliver an objective assessment of the organization's processes, controls, and adherence to compliance standards. In selecting an external consultant, it's crucial for organizations to evaluate their experience, qualifications, industry-specific knowledge, and reputation. Establishing clear expectations, defining the scope of work, and agreeing on deliverables are key steps to ensure a fruitful collaboration with a consultant. Conclusion: Harnessing the power of internal audits for organizational growth Internal audits serve as a vital instrument for organizations to scrutinize their internal controls, engage in risk management, and verify compliance with regulatory standards. The routine execution of internal audits can bolster operational efficiency, mitigate risks, and sharpen the decision-making processes. Moreover, internal audits are integral to achieving ISO certification, as they demonstrate an organization's adherence to the stringent ISO standards. Despite potential hurdles, by adhering to best practices, leveraging available tools and resources, and possibly seeking external expertise, organizations can maximize the benefits of internal audits and contribute to sustained organizational growth. CTA: To learn more about the benefits of internal audits and how they can propel your organization forward, we invite you to contact us for a comprehensive consultation. Allow us to guide you in leveraging the strategic advantages of internal audits for your organization's triumph.