Preventive controls
- Preventive controls are measures taken to prevent or mitigate hazards in food.
- The U.S. Food and Drug Administration (FDA) Food Safety Modernization Act (FSMA) requires food facilities to implement preventive controls to ensure the safety of food.
- There are four types of preventive controls:
- Process controls are measures taken to prevent or mitigate hazards during food processing.
- food allergen controls are measures taken to prevent or mitigate hazards from food allergens.
- Sanitation controls are measures taken to prevent or mitigate hazards from contamination of food contact surfaces.
- supply-chain controls are measures taken to prevent or mitigate hazards from contamination during transportation and storage.
- Preventive controls must be based on hazard analysis. A hazard analysis is a scientific evaluation of the risks posed by a food hazard. The hazard analysis must identify the potential hazards and assess the risks posed by those hazards.
- The FDA has developed resources to help food facilities develop and implement preventive controls. These resources include the Preventive Controls for Human Food rule and the Small Entity Compliance Guide.
- Preventive controls are procedures, practices, and processes that help prevent the occurrence of food safety hazards.
- The U.S. Food and Drug Administration (FDA) has developed regulations for preventive controls for human food and animal food under the Food Safety Modernization Act (FSMA).
- The FSMA final rule on preventive controls for human food requires food facilities to have a food safety plan that includes preventive controls to address food safety hazards.
- Preventive controls can be used to control physical, chemical, and biological food safety hazards.
- Physical hazards can be controlled by procedures such as cleaning and sanitation, cooking, and cooling.
- Chemical hazards can be controlled by procedures such as food allergens, food additives, and pesticide residues.
- Biological hazards can be controlled by procedures such as pathogen control, time and temperature control, and personal hygiene.
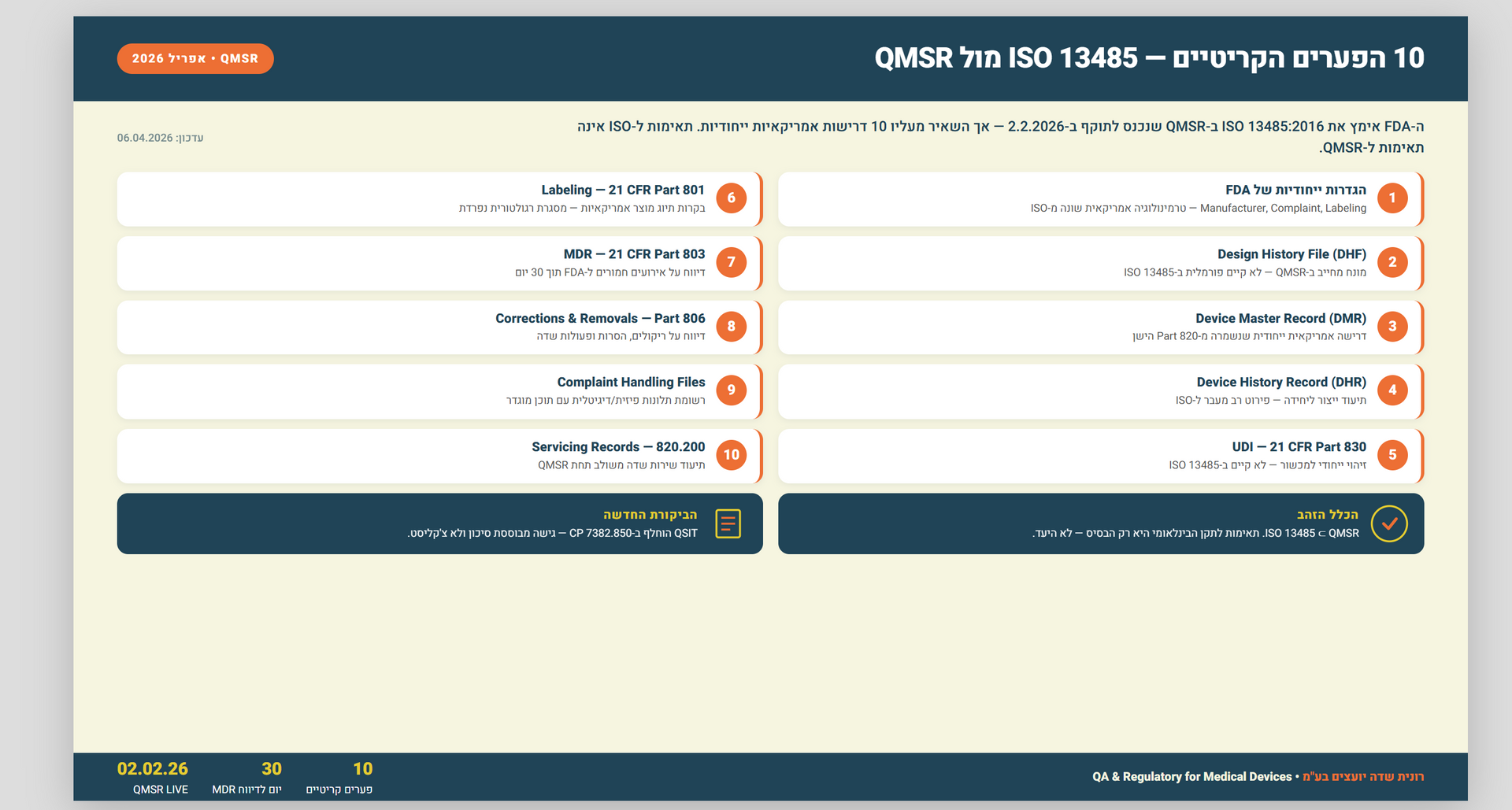
: ISO 13485 מול QMSR — מדריך השוואה מקצועי לכל הפערים בין התקן הבינלאומי לרגולציית ה-FDA החדשה שנכנסה לתוקף בפברואר 2026. עם דוגמאות ופתרונות יישום.
תשובה קצרה: QMSR של ה-FDA, שנכנס לתוקף ב-2 בפברואר 2026, מאמץ את ISO 13485:2016 בהפניה (by reference) אך מוסיף מעליו דרישות אמריקאיות ייחודיות. לכן תאימות מלאה ל-ISO 1348