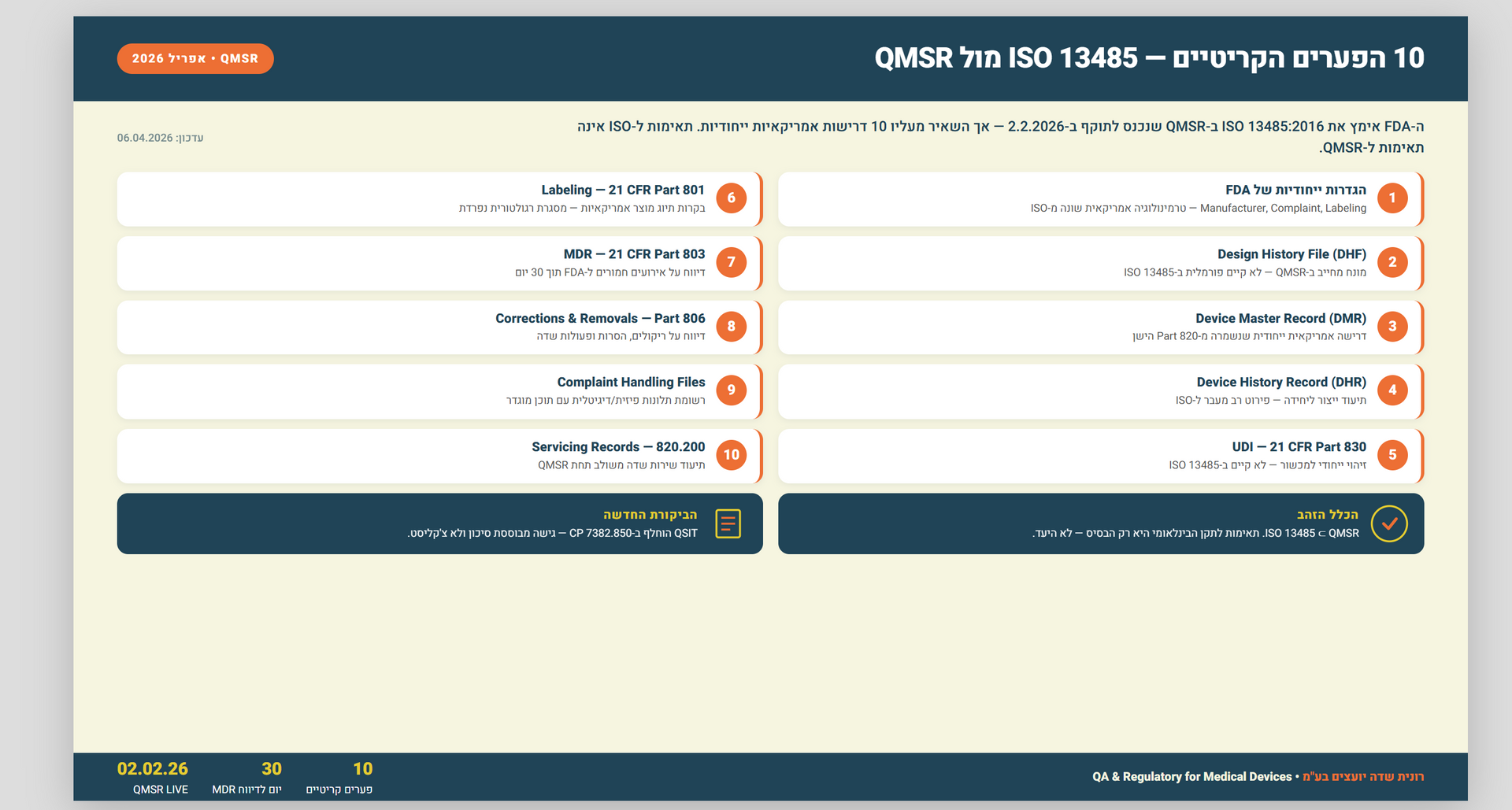
Ensuring Compliance: A Comprehensive Guide to Preparing for ISO 13485:2016 External Audit
Introduction to ISO 13485:2016
As a medical device manufacturer, ensuring compliance with industry standards is essential for the safety and well-being of patients. One such standard is ISO 13485:2016, which outlines the requirements for a quality management system (QMS) specific to the medical device industry. Compliance with this standard is mandatory for companies that sell medical devices in certain countries, including the European Union and Canada.
ISO 13485:2016 provides a framework for the development, implementation, and maintenance of a QMS that is focused on the safety and effectiveness of medical devices. The standard covers all aspects of the device life cycle, from design and development to production, distribution, and post-market activities. It is designed to ensure that medical devices consistently meet regulatory requirements and customer expectations.
Achieving compliance with ISO 13485:2016 requires a thorough understanding of the standard, as well as a commitment to continuous improvement and adherence to best practices. One important aspect of this process is the external audit, which is conducted by a certified auditor to evaluate a company's compliance with the standard. In this article, we will discuss the external audit process and provide tips and best practices for preparing for and maintaining compliance with ISO 13485:2016.
Understanding the external audit process
The external audit is a critical component of the ISO 13485:2016 compliance process. It is an independent, objective evaluation of a company's QMS by a certified auditor, who assesses whether the QMS meets the requirements of the standard. The audit is typically conducted on-site at the company's facilities and may take several days to complete, depending on the size and complexity of the organization.
During the audit, the auditor will review documentation, interview personnel, and observe processes to determine whether the QMS is effective and compliant with the standard. The auditor will also identify any nonconformities, or areas where the company is not meeting the requirements of the standard.
At the conclusion of the audit, the auditor will provide a report that outlines the findings and any nonconformities. The company will then be required to develop a corrective action plan to address the nonconformities and demonstrate compliance with the standard.
Benefits of preparing for an external audit
Preparing for an external audit can be a challenging and time-consuming process, but it offers several benefits for medical device manufacturers. First and foremost, it ensures compliance with ISO 13485:2016, which is essential for selling medical devices in certain countries. Compliance with the standard also demonstrates a commitment to quality and safety, which can enhance a company's reputation and increase customer confidence.
Preparing for an external audit can also help identify areas for improvement in the QMS. By conducting a self-assessment and developing an audit plan, companies can identify potential nonconformities and take steps to address them before the audit. This can improve the effectiveness of the QMS and reduce the likelihood of nonconformities being identified during the audit.
Finally, preparing for an external audit can help companies build a relationship with the auditor. By working collaboratively with the auditor and demonstrating a commitment to compliance, companies can establish a positive rapport that can be beneficial for future audits.
Key requirements of ISO 13485:2016
ISO 13485:2016 outlines several key requirements for a QMS specific to the medical device industry. These include:
- Management responsibility - top management must demonstrate a commitment to quality and ensure that the QMS is effectively implemented and maintained.
- Resource management - the company must provide the necessary resources, including personnel, infrastructure, and equipment, to support the QMS.
- Design and development - the company must have a documented design and development process that is risk-based and includes verification and validation activities.
- Purchasing - the company must have a process for selecting and evaluating suppliers and ensuring that purchased products meet the necessary requirements.
- Production and service provision - the company must have documented processes for production and service provision that ensure the consistent quality of the product or service.
- Monitoring and measurement - the company must have a process for monitoring and measuring the effectiveness of the QMS and taking corrective action when necessary.
- Improvement - the company must have a process for continually improving the QMS, including addressing nonconformities and implementing preventive actions.
Preparing for an external audit - tips and best practices
Preparing for an external audit requires a comprehensive approach that includes a self-assessment, audit plan development, and collaboration with the auditor. Here are some tips and best practices for each of these steps:
Conducting a self-assessment
A self-assessment is a critical first step in preparing for an external audit. It allows companies to identify potential nonconformities and take corrective action before the audit. Here are some tips for conducting a self-assessment:
- Review the ISO 13485:2016 standard and ensure that all requirements are met.
- Identify areas for improvement and develop a corrective action plan.
- Review documentation and ensure that it is accurate, up-to-date, and meets the requirements of the standard.
- Conduct internal audits to ensure that processes are effective and compliant with the standard.
Developing an audit plan
An audit plan is a roadmap for the external audit that outlines the scope, objectives, and timelines of the audit. Here are some tips for developing an audit plan:
- Identify the audit team and ensure that they have the necessary knowledge and experience to conduct the audit.
- Determine the scope of the audit and ensure that all relevant processes and departments are included.
- Develop a timeline for the audit that allows sufficient time for the auditor to review documentation, interview personnel, and observe processes.
- Ensure that all necessary resources, including personnel and equipment, are available during the audit.
Working with an auditor
Collaboration with the auditor is essential for a successful external audit. Here are some tips for working with an auditor:
- Establish a positive rapport with the auditor and demonstrate a commitment to compliance.
- Provide the auditor with access to all necessary documentation and personnel.
- Respond promptly to any requests or questions from the auditor.
- Be open and transparent about any nonconformities or areas for improvement.
Common audit findings and how to address them
Despite best efforts, it is possible that an external audit may identify nonconformities or areas for improvement. Here are some common audit findings and how to address them:
- Inadequate documentation - ensure that all documentation is accurate, up-to-date, and meets the requirements of the standard.
- Lack of training - ensure that all personnel are trained on the QMS and understand their roles and responsibilities.
- Inconsistent processes - ensure that processes are documented, followed, and regularly reviewed for effectiveness.
- Nonconforming products - ensure that all products are thoroughly evaluated and tested before release.
- Inadequate corrective action - ensure that corrective action plans are developed, implemented, and monitored for effectiveness.
Maintaining compliance after the audit
Achieving compliance with ISO 13485:2016 is an ongoing process that requires continuous improvement and adherence to best practices. Here are some tips for maintaining compliance after the audit:
- Conduct regular internal audits to identify potential nonconformities and areas for improvement.
- Review and update the QMS as necessary to ensure that it remains effective and compliant with the standard.
- Ensure that all personnel are trained on the QMS and understand their roles and responsibilities.
- Monitor and measure the effectiveness of the QMS and take corrective action when necessary.
- Maintain open communication with the auditor and address any nonconformities or areas for improvement promptly.
Conclusion - the importance of ISO 13485:2016 compliance
Ensuring compliance with ISO 13485:2016 is essential for medical device manufacturers that sell products in certain countries. Compliance with the standard demonstrates a commitment to quality and safety and can enhance a company's reputation and increase customer confidence.
Preparing for an external audit requires a comprehensive approach that includes a self-assessment, audit plan development, and collaboration with the auditor. By following best practices and addressing any nonconformities or areas for improvement, companies can achieve and maintain compliance with the standard.
Remember, the audit process is not just about achieving compliance, but also about continuously improving the effectiveness of the QMS. By staying committed to compliance and quality, medical device manufacturers can ensure the safety and well-being of patients and build a successful and sustainable business.