How To Address Document Fraud In Food Fraud Vulnerability Assessments
Document fraud can take many forms in the food industry, and it can occur at various stages of the supply chain. For example, a supplier may falsify documentation to misrepresent the origin or quality of a raw material. This could include altering the country of origin or falsifying a certificate of analysis to indicate that the material meets certain quality standards when, in fact, it does not.
In another example, a manufacturer may falsify production records to indicate that a certain number of products were produced when, in fact, they were not. This could be done to meet production targets or to satisfy customer demand, but it could result in a shortage of products, leading to customer dissatisfaction or even a potential food safety issue if the products are not produced according to established standards.
Document fraud can also occur in the transportation and distribution stages of the supply chain. For example, a distributor may alter the quantity or quality of products on a bill of lading to overstate the value of a shipment and charge a higher price to the buyer. Alternatively, a driver may falsify a delivery record to cover up the theft of a portion of the shipment.
These are just a few examples of how document fraud can occur in the food industry. It highlights the importance of conducting a thorough food fraud vulnerability assessment that includes an evaluation of document management systems to identify potential risks and vulnerabilities and develop effective countermeasures to prevent, detect, and respond to fraudulent activities.
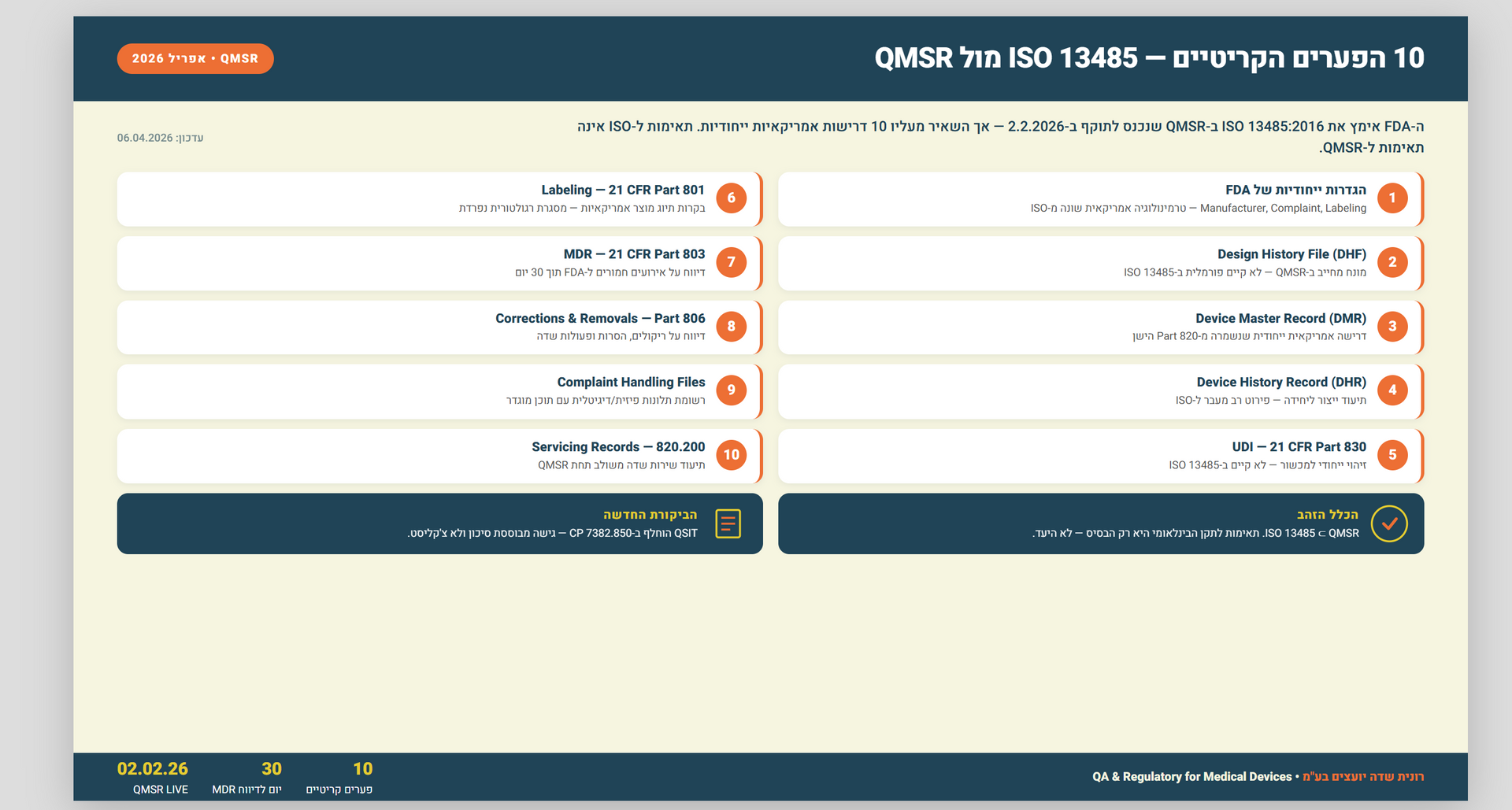
Document fraud is a common issue in the food industry, where the integrity of documentation is critical for ensuring food safety and quality. In recent years, there has been a growing concern about the risk of food fraud, including the possibility of document fraud. Document fraud can occur at various stages of the food supply chain, from raw material sourcing to manufacturing and distribution. To address this issue, it is crucial to conduct food fraud vulnerability assessments, which should include a thorough evaluation of document management systems.
Food fraud vulnerability assessments are a vital tool for identifying potential risks and vulnerabilities in the food supply chain. These assessments help organizations to identify the areas where they are most susceptible to fraud, so they can develop and implement effective countermeasures to prevent, detect, and respond to fraudulent activities. In the case of document fraud, there are several key steps that organizations can take to address the issue and protect their businesses.
- Conduct Document Audits
The first step in addressing document fraud in a food fraud vulnerability assessment is to conduct a comprehensive document audit. This audit should include an evaluation of all documents that are critical to the food supply chain, such as invoices, purchase orders, bills of lading, certificates of analysis, and other records. The audit should aim to identify any inconsistencies or irregularities in the documents that may indicate fraud, such as alterations, missing information, or false information.
- Implement Document Control Procedures
The second step in addressing document fraud is to implement document control procedures that can help prevent fraud from occurring. These procedures should include a clear set of guidelines and policies for document management, including who has access to documents, how they are stored, and how they are verified. This can help to ensure that only authorized personnel can access critical documents, and that they are not tampered with or falsified.
- Conduct Employee Training
Employee training is another critical aspect of addressing document fraud in a food fraud vulnerability assessment. Employees should be trained on the importance of document integrity, the risks of document fraud, and the procedures for document control. This training should be ongoing, with regular updates and refresher courses to ensure that employees are aware of the latest threats and countermeasures.
- Implement Technology Solutions
Technology solutions can also help to address document fraud in a food fraud vulnerability assessment. This can include the use of electronic document management systems that can track and monitor document changes, as well as the use of authentication technologies such as digital signatures and barcodes. These technologies can help to ensure that documents are not altered or falsified and can provide a record of who has accessed or modified them.
- Regularly Review and Update Procedures
Finally, it is crucial to regularly review and update document control procedures to ensure that they remain effective and up-to-date with the latest threats and countermeasures. This can include conducting regular audits, reviewing employee training programs, and evaluating technology solutions to identify any areas where improvements can be made.
In conclusion, document fraud is a serious issue in the food industry, and it is essential to address it in a food fraud vulnerability assessment. By conducting document audits, implementing document control procedures, conducting employee training, implementing technology solutions, and regularly reviewing and updating procedures, organizations can protect themselves from the risks of document fraud and ensure the integrity of their documents and the safety and quality of their food products.